When developing a cosmetic product for multiple markets, one question always comes up:
Is our formula compliant everywhere we want to sell?
At first glance, this might seem like a straightforward regulatory check.
In reality, it is one of the most complex and underestimated challenges in cosmetic product development.
Each market has its own regulatory framework, ingredient restrictions, concentration limits, and labeling requirements. A formula that is compliant in one region may require adjustments or even reformulation in another.
And as product portfolios grow, this complexity increases exponentially.
This article provides a practical and structured approach to verifying cosmetic formula compliance across multiple markets and explains how teams manage this efficiently in real-world conditions.
Why compliance differs between markets
Cosmetic regulations are not harmonized globally, even if they often share similar principles.
European Union
The EU regulatory framework is one of the most detailed, with:
- strict prohibited and restricted ingredient lists (Annex II & III)
- mandatory safety assessment (CPSR)
- detailed labeling requirements
United States (MoCRA)
The U.S. framework has evolved significantly with MoCRA:
- facility registration requirements
- product listing obligations
- increased focus on safety substantiation
However, ingredient restrictions are less centralized compared to the EU.
Canada
Canada combines elements of both approaches:
- Cosmetic Ingredient Hotlist (restrictions)
- Cosmetic Notification Form (CNF)
- labeling and bilingual requirements
👉 Key takeaway:
Compliance is not only about ingredients—it’s about how those ingredients are regulated in each market.
Step 1: Define your target markets early
One of the most common mistakes is checking compliance too late.
If markets are not clearly defined early:
- formulas may need to be adjusted post-development
- product launches may be delayed
- additional testing or documentation may be required
👉 Best practice:
Define from the beginning:
- primary markets
- secondary expansion markets
- regulatory constraints per region
Step 2: Perform a detailed ingredient screening
Each ingredient must be assessed individually.
This includes:
- prohibited status
- restricted status
- allowed conditions
For example:
An ingredient may be:
- allowed in the EU but restricted in Canada
- allowed in rinse-off but not in leave-on products
- allowed only below a certain concentration
👉 This is where most compliance issues start.
Step 3: Analyze concentration limits and use conditions
Compliance is not binary.
Many ingredients are allowed but under specific conditions:
- maximum concentration limits
- product type restrictions
- required warnings
👉 Example:
Fragrance allergens must be disclosed above specific thresholds in Canada and the EU.
Ignoring these details can lead to:
- non-compliant labeling
- regulatory rejection
- product reformulation
Step 4: Validate claims and classification
Regulatory compliance is not only about formulation—it is also about positioning.
Claims can change the regulatory category of a product.
Examples:
- “anti-aging” → cosmetic
- “anti-acne” → regulated as a drug (US/Canada)
- “SPF protection” → specific regulatory frameworks
👉 Misclassification is one of the most common hidden risks.
Step 5: Check labeling requirements across markets
Labeling requirements vary significantly:
- ingredient list (INCI format)
- warnings
- language requirements (e.g., English/French in Canada)
- mandatory information
👉 Many companies discover compliance issues at the labeling stage—not formulation.
Step 6: Ensure documentation and traceability
Regulators may request:
- safety justification
- ingredient data
- formulation history
- updates over time
Without proper documentation:
👉 even a compliant formula can become a regulatory risk.
Where companies struggle in real life
In theory, the process is clear.
In practice, challenges come from:
- data scattered across multiple tools
- lack of version control
- inconsistent ingredient naming
- manual checks across markets
As portfolios grow, this becomes unmanageable.
How companies manage formula compliance today
Today, most companies handling multiple markets do not rely solely on manual processes.
They use structured systems to:
- centralize formula data
- automate compliance checks
- monitor regulatory updates
- reduce operational risk
Because compliance is no longer a one-time validation—it is continuous.
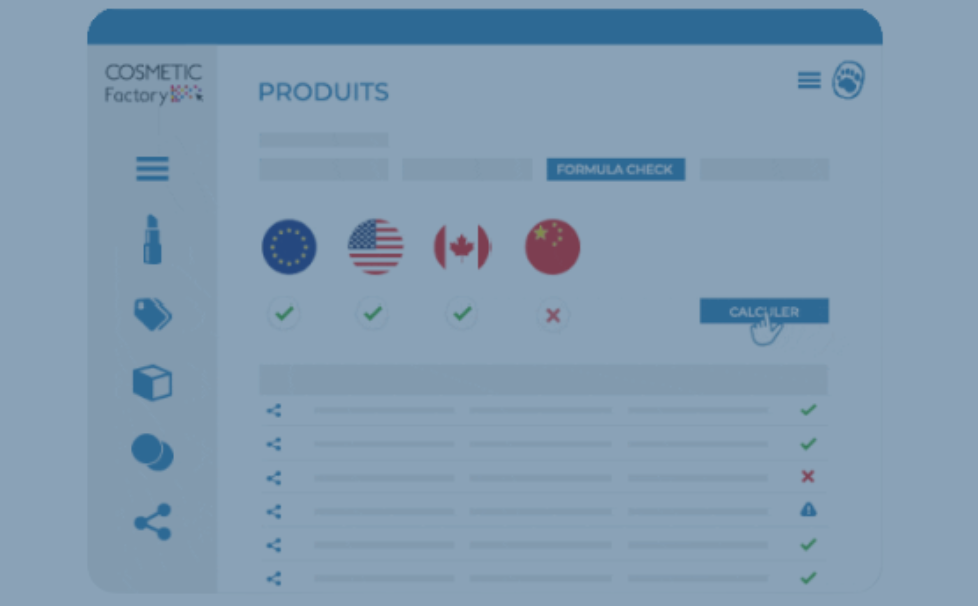
Where Formula Check makes a real difference
Within Cosmetic Factory, the Formula Check module provides a structured way to manage this complexity.
It allows teams to:
- instantly verify formula compliance across multiple markets
- identify restricted or non-compliant ingredients
- understand regulatory conditions (limits, warnings, product type)
- anticipate issues before product launch
Instead of reacting to compliance issues, teams can prevent them.
A shift from reactive to proactive compliance
The real benefit is not speed—it is control.
Companies using structured tools move from:
❌ reactive compliance (fixing issues late)
to
✔ proactive compliance (anticipating risks early)
Conclusion
Checking cosmetic formula compliance across markets is not just a regulatory task—it is a strategic capability.
Companies that manage it well:
- reduce delays
- avoid reformulation
- accelerate product launches
And most importantly, they stay in control of their regulatory environment.
See Formula Check in action
If you want to understand how teams manage multi-market formula compliance in practice, we’d be happy to show you.
When developing a cosmetic product for multiple markets, one question always comes up:
Is our formula compliant everywhere we want to sell?
At first glance, this might seem like a straightforward regulatory check.
In reality, it is one of the most complex and underestimated challenges in cosmetic product development.
Each market has its own regulatory framework, ingredient restrictions, concentration limits, and labeling requirements. A formula that is compliant in one region may require adjustments or even reformulation in another.
And as product portfolios grow, this complexity increases exponentially.
This article provides a practical and structured approach to verifying cosmetic formula compliance across multiple markets and explains how teams manage this efficiently in real-world conditions.
Why compliance differs between markets
Cosmetic regulations are not harmonized globally, even if they often share similar principles.
European Union
The EU regulatory framework is one of the most detailed, with:
- strict prohibited and restricted ingredient lists (Annex II & III)
- mandatory safety assessment (CPSR)
- detailed labeling requirements
United States (MoCRA)
The U.S. framework has evolved significantly with MoCRA:
- facility registration requirements
- product listing obligations
- increased focus on safety substantiation
However, ingredient restrictions are less centralized compared to the EU.
Canada
Canada combines elements of both approaches:
- Cosmetic Ingredient Hotlist (restrictions)
- Cosmetic Notification Form (CNF)
- labeling and bilingual requirements
👉 Key takeaway:
Compliance is not only about ingredients—it’s about how those ingredients are regulated in each market.
Step 1: Define your target markets early
One of the most common mistakes is checking compliance too late.
If markets are not clearly defined early:
- formulas may need to be adjusted post-development
- product launches may be delayed
- additional testing or documentation may be required
👉 Best practice:
Define from the beginning:
- primary markets
- secondary expansion markets
- regulatory constraints per region
Step 2: Perform a detailed ingredient screening
Each ingredient must be assessed individually.
This includes:
- prohibited status
- restricted status
- allowed conditions
For example:
An ingredient may be:
- allowed in the EU but restricted in Canada
- allowed in rinse-off but not in leave-on products
- allowed only below a certain concentration
👉 This is where most compliance issues start.
Step 3: Analyze concentration limits and use conditions
Compliance is not binary.
Many ingredients are allowed but under specific conditions:
- maximum concentration limits
- product type restrictions
- required warnings
👉 Example:
Fragrance allergens must be disclosed above specific thresholds in Canada and the EU.
Ignoring these details can lead to:
- non-compliant labeling
- regulatory rejection
- product reformulation
Step 4: Validate claims and classification
Regulatory compliance is not only about formulation—it is also about positioning.
Claims can change the regulatory category of a product.
Examples:
- “anti-aging” → cosmetic
- “anti-acne” → regulated as a drug (US/Canada)
- “SPF protection” → specific regulatory frameworks
👉 Misclassification is one of the most common hidden risks.
Step 5: Check labeling requirements across markets
Labeling requirements vary significantly:
- ingredient list (INCI format)
- warnings
- language requirements (e.g., English/French in Canada)
- mandatory information
👉 Many companies discover compliance issues at the labeling stage—not formulation.
Step 6: Ensure documentation and traceability
Regulators may request:
- safety justification
- ingredient data
- formulation history
- updates over time
Without proper documentation:
👉 even a compliant formula can become a regulatory risk.
Where companies struggle in real life
In theory, the process is clear.
In practice, challenges come from:
- data scattered across multiple tools
- lack of version control
- inconsistent ingredient naming
- manual checks across markets
As portfolios grow, this becomes unmanageable.
How companies manage formula compliance today
Today, most companies handling multiple markets do not rely solely on manual processes.
They use structured systems to:
- centralize formula data
- automate compliance checks
- monitor regulatory updates
- reduce operational risk
Because compliance is no longer a one-time validation—it is continuous.
Where Formula Check makes a real difference
Within Cosmetic Factory, the Formula Check module provides a structured way to manage this complexity.
It allows teams to:
- instantly verify formula compliance across multiple markets
- identify restricted or non-compliant ingredients
- understand regulatory conditions (limits, warnings, product type)
- anticipate issues before product launch
Instead of reacting to compliance issues, teams can prevent them.
A shift from reactive to proactive compliance
The real benefit is not speed—it is control.
Companies using structured tools move from:
❌ reactive compliance (fixing issues late)
to
✔ proactive compliance (anticipating risks early)
Conclusion
Checking cosmetic formula compliance across markets is not just a regulatory task—it is a strategic capability.
Companies that manage it well:
- reduce delays
- avoid reformulation
- accelerate product launches
And most importantly, they stay in control of their regulatory environment.
See Formula Check in action
If you want to understand how teams manage multi-market formula compliance in practice, we’d be happy to show you.







