
When talking about IFRA, the first thing to come to our mind is of course perfumes, however, more than perfume, IFRA impacts several categories of products of which, the cosmetic products but also, homecare products, such as diffuser, candles or detergents. The IFRA standards are frequently updated, and as of now, 51 standards have been published on June 30th, 2023. (All the contents were published on July 3rd, 2023 on their website)
What ingredients are involved?
Following the publication of the new IFRA standards, 11 perfume ingredients have their restrictions changed, and 48 perfume ingredients are added in this new amendment.
New standards:
- 1 new IFRA Restriction Specification Standard (Allyl-3-cyclohexylpropionate)
- 32 new IFRA Restriction Standards to control potential dermal sensitization effects for which the systemic toxicity endpoints have also been evaluated (like Isobutyl cinnamate, cis-3-Nonenyl acetate and Ethyl and methyl furaneol).
- 11 new IFRA Restriction Standards to control potential dermal sensitization effects based solely on QRA2 (including carvyl acetate, methyl lavender ketone and 3-Octen-2-one).
- 2 new IFRA Restriction Standards, for which Risk Management is based on, Threshold of Toxicological Concern (TTC) for Methoxycyclododecane and 7-Methoxy-3,7-dimethyloct-1-ene.
- 1 new IFRA Restriction Standard due to potential of depigmentation for Cresols.
- 1 new IFRA Prohibition Standard due to potential genotoxicity effects for 3-Acetyl-2,5-dimethylfuran.
Revised standard:
- 7 Revised IFRA Restriction Standards to control potential dermal sensitization effects for which the systemic toxicity endpoints have now also been evaluated.
- 1 Revised IFRA Restriction Standard based on new dermal sensitization data.
- 1 Revised IFRA Restriction Standard based on phototoxicity and systemic toxicity (and 1 updated Standard with regard to MAC in Category 6).
- 2 Revised IFRA Restriction Standards based on systemic toxicity.
- Minor updates to existing Standards and format change.
Good to know: Consequence of the revision of one standard
One of the consequences of IFRA 51, is the new approach concerning phototoxic ingredients. This approach now includes setting limits for rinse-off products in Categories 7A, 9, and 10A. Specifically, this change has been implemented for Methyl-N-Methyl Anthranilate (MNMA).
Furthermore, the Fragrance Safety Expert Group has determined that products in Category 6 can now be classified as rinse-off products in terms of phototoxicity. Consequently, the allowed concentration of Methyl-N-Methyl Anthranilate in Category 6 products has increased from 0.1% to 0.5%, as compared to the previous consultation.
What about the transition period ?
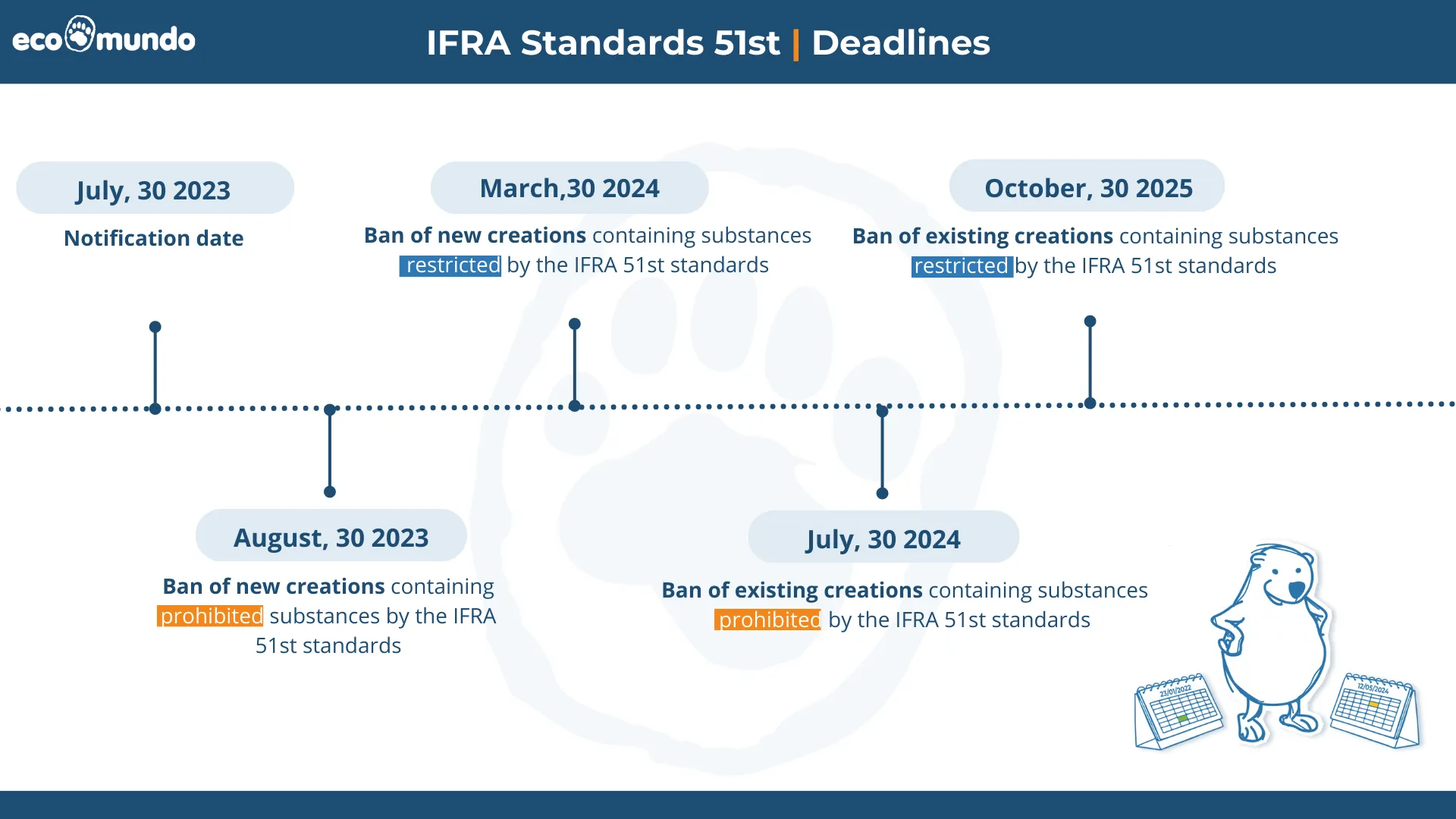
A grace period has been established based on four cases scenario, depending on the related product's current presence on the market and whether the fragrance is prohibited or restricted :
- If the fragrance is created after the 51st amendment notification and your raw material contains a (future) prohibited ingredient, you will have 2 months to be compliant with this new amendment. (30, August 2023)
- If the fragrance is created after the 51st amendment notification and your perfume does not contain prohibited ingredients but contains restricted ingredients or ingredients with specification, you will have 9 months. (30, March 2024)
- If the fragrance is already on the market before the 51st amendment notification and your raw material contains a (future) prohibited ingredient, you will have 13 months to be compliant with this new amendment. (30, July 2024)
- If the fragrance is already on the market before the 51st amendment notification and your perfume does not contain prohibited ingredients but contains restricted ingredients or ingredients with specification, you will have 28 months. (30, October 2025)
How to stay compliant?
Now that the 51st IFRA standards have been published, you have to ensure your compliance, by collecting the updated IFRA certificate from your suppliers for the fragrance concentrates, flavors and essential oils of your products.
Be aware that these changes can directly impact your finished products. Keep in mind that IFRA standards do not substitute for national regulations or safety assessment!
For example in Europe, there can be discrepancies between the IFRA standards and the Cosmetic Product Regulations EC 1223/2009, so make sure you comply with both.
If you have any doubts you can contact EcoMundo to figure out how to be IFRA compliant depending on your targeted regions !
When talking about IFRA, the first thing to come to our mind is of course perfumes, however, more than perfume, IFRA impacts several categories of products of which, the cosmetic products but also, homecare products, such as diffuser, candles or detergents. The IFRA standards are frequently updated, and as of now, 51 standards have been published on June 30th, 2023. (All the contents were published on July 3rd, 2023 on their website)
What ingredients are involved?
Following the publication of the new IFRA standards, 11 perfume ingredients have their restrictions changed, and 48 perfume ingredients are added in this new amendment.
New standards:
- 1 new IFRA Restriction Specification Standard (Allyl-3-cyclohexylpropionate)
- 32 new IFRA Restriction Standards to control potential dermal sensitization effects for which the systemic toxicity endpoints have also been evaluated (like Isobutyl cinnamate, cis-3-Nonenyl acetate and Ethyl and methyl furaneol).
- 11 new IFRA Restriction Standards to control potential dermal sensitization effects based solely on QRA2 (including carvyl acetate, methyl lavender ketone and 3-Octen-2-one).
- 2 new IFRA Restriction Standards, for which Risk Management is based on, Threshold of Toxicological Concern (TTC) for Methoxycyclododecane and 7-Methoxy-3,7-dimethyloct-1-ene.
- 1 new IFRA Restriction Standard due to potential of depigmentation for Cresols.
- 1 new IFRA Prohibition Standard due to potential genotoxicity effects for 3-Acetyl-2,5-dimethylfuran.
Revised standard:
- 7 Revised IFRA Restriction Standards to control potential dermal sensitization effects for which the systemic toxicity endpoints have now also been evaluated.
- 1 Revised IFRA Restriction Standard based on new dermal sensitization data.
- 1 Revised IFRA Restriction Standard based on phototoxicity and systemic toxicity (and 1 updated Standard with regard to MAC in Category 6).
- 2 Revised IFRA Restriction Standards based on systemic toxicity.
- Minor updates to existing Standards and format change.
Good to know: Consequence of the revision of one standard
One of the consequences of IFRA 51, is the new approach concerning phototoxic ingredients. This approach now includes setting limits for rinse-off products in Categories 7A, 9, and 10A. Specifically, this change has been implemented for Methyl-N-Methyl Anthranilate (MNMA).
Furthermore, the Fragrance Safety Expert Group has determined that products in Category 6 can now be classified as rinse-off products in terms of phototoxicity. Consequently, the allowed concentration of Methyl-N-Methyl Anthranilate in Category 6 products has increased from 0.1% to 0.5%, as compared to the previous consultation.
What about the transition period ?
A grace period has been established based on four cases scenario, depending on the related product's current presence on the market and whether the fragrance is prohibited or restricted :
- If the fragrance is created after the 51st amendment notification and your raw material contains a (future) prohibited ingredient, you will have 2 months to be compliant with this new amendment. (30, August 2023)
- If the fragrance is created after the 51st amendment notification and your perfume does not contain prohibited ingredients but contains restricted ingredients or ingredients with specification, you will have 9 months. (30, March 2024)
- If the fragrance is already on the market before the 51st amendment notification and your raw material contains a (future) prohibited ingredient, you will have 13 months to be compliant with this new amendment. (30, July 2024)
- If the fragrance is already on the market before the 51st amendment notification and your perfume does not contain prohibited ingredients but contains restricted ingredients or ingredients with specification, you will have 28 months. (30, October 2025)

How to stay compliant?
Now that the 51st IFRA standards have been published, you have to ensure your compliance, by collecting the updated IFRA certificate from your suppliers for the fragrance concentrates, flavors and essential oils of your products.
Be aware that these changes can directly impact your finished products. Keep in mind that IFRA standards do not substitute for national regulations or safety assessment!
For example in Europe, there can be discrepancies between the IFRA standards and the Cosmetic Product Regulations EC 1223/2009, so make sure you comply with both.
If you have any doubts you can contact EcoMundo to figure out how to be IFRA compliant depending on your targeted regions !







