Preamble:
Do you know what the CLP regulation is? Do you know the changes compare to the old classification system DSD / DPD? If in doubt read the article "CLP prerequisite ".
Are you affected by the CLP transition? Take the test!
Which information must appear on your label?
Article 17 of the CLP Regulation EC No 1272/2008 sets out the requirements to correctly complete sections 1 and 2 of your Safety Data Sheet (MSDS).
In order to be compliant with the CLP Regulation, for a substance or mixture classified as hazardous and/or contained in a packaging, the label must state:
- 1- Name, address and phone number of the supplier(s)
- 2- Nominal quantity of the substance or mixture in the package where this is being made available to the general public, unless this quantity is specified elsewhere on the package
- 3- Product identifiers: name and identity of the substance contributing to the classification of a mixture (see Article 18 CLP) -where applicable
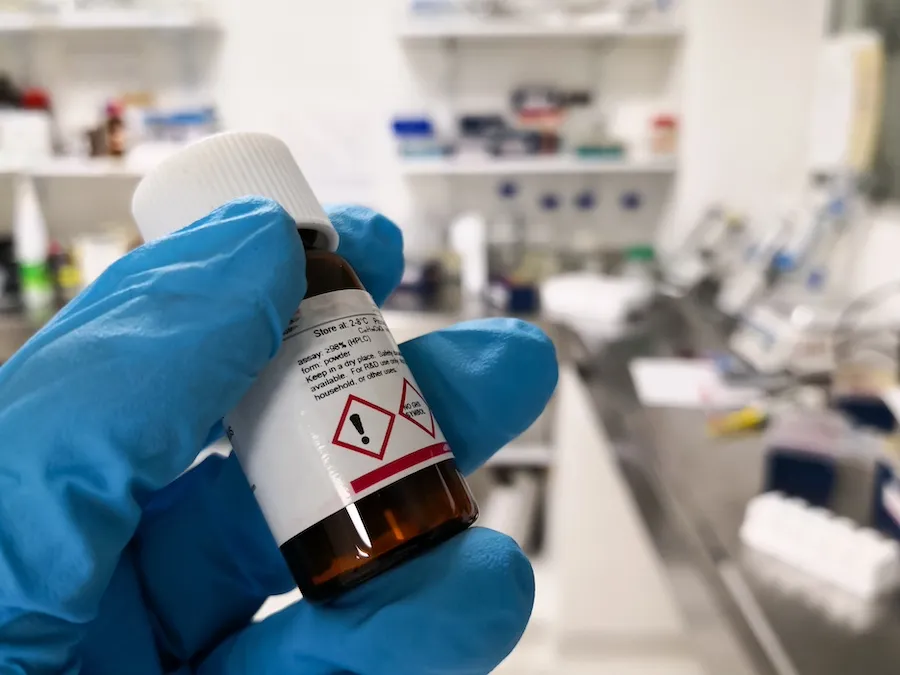
- 4- Hazard pictograms in accordance with Article 19 � where applicable
- 5- Relevant signal word in accordance with Article 20 - Where applicable
- 6- Appropriate precautionary statements in accordance with Article 22 � Where applicable
- 7- A section for supplemental information in accordance with Article 25 � Where applicable
NB: All information has to be supplied in in official languages of the Member States States in which the products are placed on the market.
Pictograms and hazard statements: what obligations for the labels?
General rules for use of the pictograms.
A hazard pictogram is according ECHA : "an image on a label includes a warning symbol and specific colours intended to provide information about the damage of a particular substance or mixture can cause to our health or the environment. "
These symbols change formalism from the DSD / DPD to CLP formalism system under the Globally Harmonized System (GHS). New pictograms have the form of a red diamond on white background with a black symbol.
Distinction between H and P phrases
The dangers are divided into 28 classes relating to physical hazards (16), dangers to health (10) or the environment (2).
The CLP regulation replaces the risk phrases (R phrase) by hazard statements (H phrase). There is no correspondence between the old R-phrase and H-phrase, although some H phrases are identical to the old R-phrase.
A hazard statement is "a phrase assigned to a hazard class or hazard category that describes the nature of the hazards of a chemical product and, where appropriate, the degree of hazard." The letter "H" stands for "Hazard = risk". 3-digit codes are added to sentences to define the type of danger.
The CLP regulation implements safety advice (P-phrase). A limited number of precautionary statements must appear on the label and are selected from among those associated with the hazard categories of the product, taking into account the uses of this product. Like H phrase, P phrase have a 3-digit code used to define the preventive measures to be implemented.
The P phrase differ from S phrase, nevertheless those sentences fulfill a similar role.
Difference between the words "warning" and "danger"
On labels you need to add the mention "warning" to the less severe hazard, or the mention "danger" for more severe hazards. When the label has the signal word "danger", it cannot contain the signal word "attention" - (Article 20).
Application of labels
Regarding the order of appearance on the label (Article 32), pictogram, signal word, H-phrase, P-phrase, shall be located together on the label. When the classification of a substance or mixture would result in more than one hazard pictogram on the label, the principle of precedence set out in Article 26 shall be applied in order to reduce the number of hazard pictograms.
As a supplier, you are free to determine the order (Article 28) of appearance of H and P phrases; Nevertheless, the H-phrase shall be grouped together and the P- phrase together. They must be classified by languages if your labels are multilingual.
NB: The label does not have more than six precautionary statements unless if this is necessary to show the nature and severity of hazards (Article 28)..
Article 31 defines the general rules for the application of labels. Paragraph 5 states that �a label shall not be required when the label elements referred to article 17(1) are shown clearly on the packaging itself. In such cases, the requirements of this Chapter are applicable to a label and shall be applied to the information shown on the packaging�.
What about EUH information? - Supplemental information
The CLP Regulation includes criteria for classification and labeling rules arising Globally Harmonized System (GHS). Nevertheless, the Regulation includes EUH statements that are not part of the GHS. They correspond to the "supplemental risk phrases" from DPD / DSD regulation.
These statements appear in section 2 of the SDS (supplemental l information) and provide additional information on hazards, including dangerous mixtures with physical properties or specific danger to health.
Packaging rules
The general rules for affixing the labels are defined in article 31. You must ensure that the labels are firmly affixed to one or more surfaces of the packaging immediately containing the substance or mixture and shall be readable horizontally when the package is set down normally. The colour and presentation of any label shall be such that the hazard pictogram stands out clearly, and the elements referred in Article 17(1) shall be clearly and indelibly marked.
In labeling, exception is made when the labeling elements referred in Article 17 � 1 clearly appear on the packaging itself. Nevertheless, you must apply the rules mentioned above on the packaging.
NB: The label must cover at least 1/15 of the minimum area of the packaging and the minimum area of each hazard pictogram is 1 cm_, in accordance with Annex I, section 1.2.1.
Good to know: Specificities of CLP regulation for certain types of products
Biocidal products
Biocidal products have a specific regulation (EU No. 528/2012). Biocidal products must be indicated in section 15 of the Safety Data Sheet.
They must be labeled in accordance with the decision of authorization issued by the competent organization; in France, it�s the "Direction G�n�rale de la Pr�vention des risques" under the Ministry of Ecology, Sustainable Development and the Energy. This organisation shall issue permits on the basis of an evaluation of the National Agency for Sanitary Safety of Food Environment and Labour (ANSES).
Thus the identity of the biocidal (s) active(s) substance (s) contained in the product must appear on the label of the biocidal product. "The indication is to enable unequivocally identification of substances". The biocidal active substance concentration should be included in metric units or percentage unit. The presence of nanomaterials should also appear on the label.
The sentence 'Read attached instructions before use "must be mentioned if a manual is provided. The batch designation, preparation, and the expiry date under normal storage conditions are required.
Information to be included on the label for biocidal products:
- Time required for emergence of the biocidal effect
- Duration of action
- Interval to be observed between applications of the biocidal product or between application and next use of the product, material or surface that has been treated or the further access by human or animals the area use treated with a biocidal product
- Particulars concerning decontamination means and measures
- Measure and duration of necessary ventilation time of treated areas
Finally, the type of preparation (GIFAP codes), user type, the side-effects including indirect side-effects, first aid instructions and instructions for safe disposal of the biocidal product and its packaging are required. The sentence 'Read attached instructions before use "must be mentioned if a manual is provided.
NB: Any advertisement for a biocidal product is accompanied by the following warning: "Use biocides safely. Before using, read the label and product information." These warnings are clearly distinguishable from the advertising. (Article R522-39 of environmental code)
Articles treated with a biocidal product
According to the definition in Article 3 (1 (I)), a treated article is a substance, mixture or article (under REACH), treated with or intentionally incorporating biocides.
For example: a pillow treated against mites is considered as a treaty article. In contrast, an impregnated mosquito net treated with an insecticide is a biocidal product.
Labelling is required if a claim is made on the biocidal properties of the article or if the conditions of approval of the substance required. The information must be provided in the national language of the country where the item is placed on the market. You should therefore mention that the article incorporates biocidal and conferred property, the name of the active substance and any nanomaterial. Do not forget to mention the relevant instructions to protect humans and environment, as required.
Apart from the above cases all instructions of relevant treatment-related use to protect humans and the environment, as required must appear on the label (Section 15 SDS)
NB: On request of a consumer, obligation for providers of treated articles to provide, within 45 days and free of charge, information on the biocidal treatment.
Detergents
The detergents are regulated by the detergents regulation EC No. 648/2004. Thus specific information must appear on the label if they are intended for the general public.
You have to mention (section 15 SDS) a list of all the components listed in ascending order of their concentration and the address of the website where the consumer can obtain the full list of ingredients. Allergenic substances must appear clearly.
Paints and varnishes
Directive 2004/42 EC on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain paints and varnishes and vehicle refinishing products, establish special provisions for labels of these products.
Indeed, you must specify the product subcategory and the limit values for the content of Volatile Organic Compound (VOC), expressed in grams per liter, included in Annex II. The maximum VOC content of the ready to use product in g / L must also appear on the label.
Specificity for sprays and aerosols
Decree No. 2014-840 of 24 July 2014 giving effect to european Directive 2013/10 / EU on "aerosol dispensers EC No 1272/2008."
You must include on your aerosols and sprays labels (whatever its content):
- Hazard statement H229 "Pressurized container: may burst under the effect of heat"
- P210 and P251 safety recommendations
- P410 and P412 safety recommendations
- The precautionary statement P102 if the product is intended for the general public
- Any additional precaution of use informing consumers about the specific dangers of the product; If the aerosol dispenser is accompanied by separate instructions for use, the latter shall take the precautions above.
- When the aerosol is classified as "flammable" the mention "warning" should appear
- When the aerosol is classified as "extremely flammable" the mention "danger" should appear
NB: At 50 � C, the volume of the existing liquid phase does not exceed 90% of the net capacity.
Preamble:
Do you know what the CLP regulation is? Do you know the changes compare to the old classification system DSD / DPD? If in doubt read the article "CLP prerequisite ".
Are you affected by the CLP transition? Take the test!
Which information must appear on your label?
Article 17 of the CLP Regulation EC No 1272/2008 sets out the requirements to correctly complete sections 1 and 2 of your Safety Data Sheet (MSDS).
In order to be compliant with the CLP Regulation, for a substance or mixture classified as hazardous and/or contained in a packaging, the label must state:
- 1- Name, address and phone number of the supplier(s)
- 2- Nominal quantity of the substance or mixture in the package where this is being made available to the general public, unless this quantity is specified elsewhere on the package
- 3- Product identifiers: name and identity of the substance contributing to the classification of a mixture (see Article 18 CLP) -where applicable
- 4- Hazard pictograms in accordance with Article 19 � where applicable
- 5- Relevant signal word in accordance with Article 20 - Where applicable
- 6- Appropriate precautionary statements in accordance with Article 22 � Where applicable
- 7- A section for supplemental information in accordance with Article 25 � Where applicable
NB: All information has to be supplied in in official languages of the Member States States in which the products are placed on the market.
Pictograms and hazard statements: what obligations for the labels?
General rules for use of the pictograms.
A hazard pictogram is according ECHA : "an image on a label includes a warning symbol and specific colours intended to provide information about the damage of a particular substance or mixture can cause to our health or the environment. "
These symbols change formalism from the DSD / DPD to CLP formalism system under the Globally Harmonized System (GHS). New pictograms have the form of a red diamond on white background with a black symbol.
Distinction between H and P phrases
The dangers are divided into 28 classes relating to physical hazards (16), dangers to health (10) or the environment (2).
The CLP regulation replaces the risk phrases (R phrase) by hazard statements (H phrase). There is no correspondence between the old R-phrase and H-phrase, although some H phrases are identical to the old R-phrase.
A hazard statement is "a phrase assigned to a hazard class or hazard category that describes the nature of the hazards of a chemical product and, where appropriate, the degree of hazard." The letter "H" stands for "Hazard = risk". 3-digit codes are added to sentences to define the type of danger.
The CLP regulation implements safety advice (P-phrase). A limited number of precautionary statements must appear on the label and are selected from among those associated with the hazard categories of the product, taking into account the uses of this product. Like H phrase, P phrase have a 3-digit code used to define the preventive measures to be implemented.
The P phrase differ from S phrase, nevertheless those sentences fulfill a similar role.
Difference between the words "warning" and "danger"
On labels you need to add the mention "warning" to the less severe hazard, or the mention "danger" for more severe hazards. When the label has the signal word "danger", it cannot contain the signal word "attention" - (Article 20).
Application of labels
Regarding the order of appearance on the label (Article 32), pictogram, signal word, H-phrase, P-phrase, shall be located together on the label. When the classification of a substance or mixture would result in more than one hazard pictogram on the label, the principle of precedence set out in Article 26 shall be applied in order to reduce the number of hazard pictograms.
As a supplier, you are free to determine the order (Article 28) of appearance of H and P phrases; Nevertheless, the H-phrase shall be grouped together and the P- phrase together. They must be classified by languages if your labels are multilingual.
NB: The label does not have more than six precautionary statements unless if this is necessary to show the nature and severity of hazards (Article 28)..
Article 31 defines the general rules for the application of labels. Paragraph 5 states that �a label shall not be required when the label elements referred to article 17(1) are shown clearly on the packaging itself. In such cases, the requirements of this Chapter are applicable to a label and shall be applied to the information shown on the packaging�.
What about EUH information? - Supplemental information
The CLP Regulation includes criteria for classification and labeling rules arising Globally Harmonized System (GHS). Nevertheless, the Regulation includes EUH statements that are not part of the GHS. They correspond to the "supplemental risk phrases" from DPD / DSD regulation.
These statements appear in section 2 of the SDS (supplemental l information) and provide additional information on hazards, including dangerous mixtures with physical properties or specific danger to health.
Packaging rules
The general rules for affixing the labels are defined in article 31. You must ensure that the labels are firmly affixed to one or more surfaces of the packaging immediately containing the substance or mixture and shall be readable horizontally when the package is set down normally. The colour and presentation of any label shall be such that the hazard pictogram stands out clearly, and the elements referred in Article 17(1) shall be clearly and indelibly marked.
In labeling, exception is made when the labeling elements referred in Article 17 � 1 clearly appear on the packaging itself. Nevertheless, you must apply the rules mentioned above on the packaging.
NB: The label must cover at least 1/15 of the minimum area of the packaging and the minimum area of each hazard pictogram is 1 cm_, in accordance with Annex I, section 1.2.1.
Good to know: Specificities of CLP regulation for certain types of products
Biocidal products
Biocidal products have a specific regulation (EU No. 528/2012). Biocidal products must be indicated in section 15 of the Safety Data Sheet.
They must be labeled in accordance with the decision of authorization issued by the competent organization; in France, it�s the "Direction G�n�rale de la Pr�vention des risques" under the Ministry of Ecology, Sustainable Development and the Energy. This organisation shall issue permits on the basis of an evaluation of the National Agency for Sanitary Safety of Food Environment and Labour (ANSES).
Thus the identity of the biocidal (s) active(s) substance (s) contained in the product must appear on the label of the biocidal product. "The indication is to enable unequivocally identification of substances". The biocidal active substance concentration should be included in metric units or percentage unit. The presence of nanomaterials should also appear on the label.
The sentence 'Read attached instructions before use "must be mentioned if a manual is provided. The batch designation, preparation, and the expiry date under normal storage conditions are required.
Information to be included on the label for biocidal products:
- Time required for emergence of the biocidal effect
- Duration of action
- Interval to be observed between applications of the biocidal product or between application and next use of the product, material or surface that has been treated or the further access by human or animals the area use treated with a biocidal product
- Particulars concerning decontamination means and measures
- Measure and duration of necessary ventilation time of treated areas
Finally, the type of preparation (GIFAP codes), user type, the side-effects including indirect side-effects, first aid instructions and instructions for safe disposal of the biocidal product and its packaging are required. The sentence 'Read attached instructions before use "must be mentioned if a manual is provided.
NB: Any advertisement for a biocidal product is accompanied by the following warning: "Use biocides safely. Before using, read the label and product information." These warnings are clearly distinguishable from the advertising. (Article R522-39 of environmental code)
Articles treated with a biocidal product
According to the definition in Article 3 (1 (I)), a treated article is a substance, mixture or article (under REACH), treated with or intentionally incorporating biocides.
For example: a pillow treated against mites is considered as a treaty article. In contrast, an impregnated mosquito net treated with an insecticide is a biocidal product.
Labelling is required if a claim is made on the biocidal properties of the article or if the conditions of approval of the substance required. The information must be provided in the national language of the country where the item is placed on the market. You should therefore mention that the article incorporates biocidal and conferred property, the name of the active substance and any nanomaterial. Do not forget to mention the relevant instructions to protect humans and environment, as required.
Apart from the above cases all instructions of relevant treatment-related use to protect humans and the environment, as required must appear on the label (Section 15 SDS)
NB: On request of a consumer, obligation for providers of treated articles to provide, within 45 days and free of charge, information on the biocidal treatment.
Detergents
The detergents are regulated by the detergents regulation EC No. 648/2004. Thus specific information must appear on the label if they are intended for the general public.
You have to mention (section 15 SDS) a list of all the components listed in ascending order of their concentration and the address of the website where the consumer can obtain the full list of ingredients. Allergenic substances must appear clearly.
Paints and varnishes
Directive 2004/42 EC on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain paints and varnishes and vehicle refinishing products, establish special provisions for labels of these products.
Indeed, you must specify the product subcategory and the limit values for the content of Volatile Organic Compound (VOC), expressed in grams per liter, included in Annex II. The maximum VOC content of the ready to use product in g / L must also appear on the label.
Specificity for sprays and aerosols
Decree No. 2014-840 of 24 July 2014 giving effect to european Directive 2013/10 / EU on "aerosol dispensers EC No 1272/2008."
You must include on your aerosols and sprays labels (whatever its content):
- Hazard statement H229 "Pressurized container: may burst under the effect of heat"
- P210 and P251 safety recommendations
- P410 and P412 safety recommendations
- The precautionary statement P102 if the product is intended for the general public
- Any additional precaution of use informing consumers about the specific dangers of the product; If the aerosol dispenser is accompanied by separate instructions for use, the latter shall take the precautions above.
- When the aerosol is classified as "flammable" the mention "warning" should appear
- When the aerosol is classified as "extremely flammable" the mention "danger" should appear
NB: At 50 � C, the volume of the existing liquid phase does not exceed 90% of the net capacity.